
This may seem counterintuitive but it is the fact. This is because in going down a column you are jumping up to the next higher main energy level (n) and each energy level is further out from the nucleus - that is, a bigger atomic radius.Ītoms get smaller as you go across a row from left to right. so chopping that to 50-300 pm isn't a big deal.Ītoms get bigger as you go down a column on the periodic table. Oh well, you should just wrap your head around the general range of all atomic sizes the extremes are 31 pm and 343 pm. Sometimes we just do a generalized bit of rounding as well and say things like atoms range from about 50 pm to 300 pm which is more of a 6:1 ratio. So that is roughly a 10:1 ratio of largest to smallest. Which atom is the largest? That would be cesium, Cs, which comes in with a radius of 343 pm. Yeah, He is even smaller than hydrogen, H, which is 53 pm. 
The smallest atom on the periodic table is helium, He, and has a radius of 31 pm. All our trends describe the trend in two directions on the periodic table: 1) across a row, and 2) up and down a column. 
A trend is generally "it gets bigger" or "it gets smaller" sort of thing. Um, on explains why you see the atomic trends that we see.We use the periodic table to help us recognize certain trends of physical and chemical properties of the elements. So now instead of Shelby from here to here, with Shelby from here, maybe Children made from here to like here, right, because now all these electrons are coming closer to the nucleus because there are so many more electrons, center and even more positive nucleus. And so this extra attraction overpasses or surpasses shielding the facts of these electrons and it basically causing the electrons to come closer to the nucleus. But because there are war electrons and because the nucleus is even more positively charged that it was previously there is an increased attraction between the valence shells and the nucleus. So you have the same line of electrons having a shielding effect. So what this does is you have the same one of electrons shielding, right. So as you go, um, across the period, dreading more protons in the nucleus and you were adding more electrons to the Valance sexual to outer, outermost shell sexual. And it was also adding more profiles, Right? No sighting. So as you go, um, as you travel right as your trouble across a period on the periodic table, you're basically adding more balanced electrons. Valence electrons to the outer shell and you see a different pattern emerge. But the movie you added more protons More violence in the drug war. Night, please, of their colleague ratings. You know, they are especially susceptible to shielding from other layers of electrons, so they kind of just hang out kind of far away from the nucleus. Electrons were kind of, you know, far away from the office. So let's think about what Adam's on this side of periodic table look like, right? They only have one dance electron one or two. Well, this reason a little bit more complicated, Unfortunately. Now, why does atomic Radius increase as we go? Um, this way as we go, um, from the right side till outside. Like it previously was gonna be here to here. Yeah, And as you have warm or energy levels, it stands to reason that the atomic radius will decrease from here to here. You're gonna be adding more and more energy levels around. Well, if you look at a group, you're increasing energy levels, so appear you might only have one energy level. Let's let's try understand why, As you go down a group, the atomic radius increases. Why is it that these trans exists? Because it's not enough to just know that if you know why, Baker So let's start with the easier one. The periodic table are the largest and elements kind of in this region, the periodic table of the smallest. Decreases right, So goes to show the elements in this region. Atomic Radius increases Black White issue go across a period from the period of table atomic radius. So as you go down a column in the narrative, unless you go down a group, you will notice. 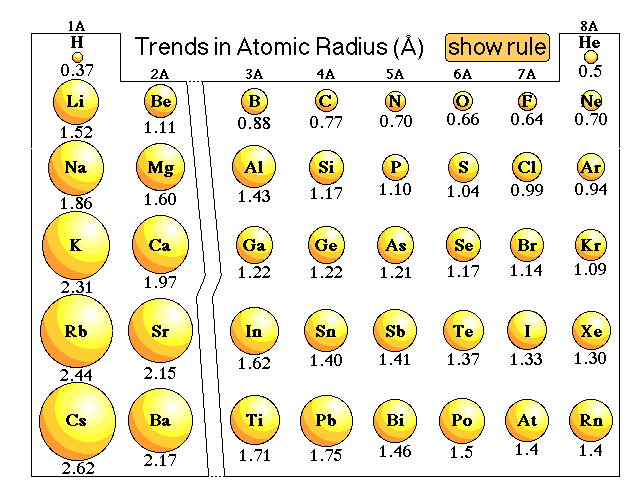
That's what is known as a top Brady's so that we had squared away one of the trends that we see. So that's the distance from nucleus to Diatta, um, lateral restaurants. So on the schematic it would be 10 of the nucleus two the peripheral valence electrons. So Tommy Radius is basically the distance between the center of the nucleus. So before we can really look at the trends of atomic radius we first needed to find what Atomic radius is. So this video is gonna take a look at the trends and atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
